Food and beverage packaging takes many shapes and forms in today’s marketplace. To maintain grocery shelf-life and flavor quality, sophisticated packaging equipment processes and assembles these products, having evolved away from traditional bottling and canning methods.
Systems of quality control in the food and beverage industry have become more sophisticated as well as companies have come under the FDA’s Hazard Analysis and Critical Control Points (HACCP) guidelines. They call for food companies to develop quality monitoring procedures at critical control production points. Furthermore, operators must identify when the process fails to meet established limits and take corrective actions. Lastly, they must maintain effective recordkeeping of food safety monitoring to document various HACCP-mandated processes.
Aseptic Packaging
Aseptic packaging technology uses both hydrogen peroxide and heat to achieve sterility, allowing food producers to distribute products through ambient temperature channels. FDA approved these processes in January 1981 in response to a petition by Tetra Pak. Within months, some of the largest U.S. beverage producers (juice, dairy, etc.) began employing aseptic packaging sterilization procedures. Although new to the U.S. at that time, the technology’s origin can be traced to Sweden’s Tetra Brik packaging introduced in 1963.

Extended Shelf Life
The chilled food and beverage segment of the market has boomed since the 1980s as packaging engineers have employed variations on the “aseptic theme” to produce systems that prolong shelf life beyond that of traditional pasteurized products (hence extended shelf-life or ESL). Although ESL processes apply a heat/time regimen that experts regard as a sterilization process, ESL packaging operations do not necessarily sterilize the packages or enclosures. As a result, distributions send all ESL products through refrigerated channels. Examples of ESL products offered in the refrigerated section of the grocery store are orange juice (not from concentrate), flavored milk, coffee creamers, and puddings.
General Protocols Used in Packaging Systems
Packaging vendors apply the same basic microbiological and engineering principles to design their food safety monitoring equipment. This holds true regardless of the food packaging system under consideration. The product type—high acid or low acid (pH) —dictates the precise details of the processes. The general procedures used to sterilize either the product or packaging systems before any product or package enters the system involve:
- steam
- steam plus hydrogen peroxide
- hydrogen peroxide
- peracetic acid
- other chemical treatments
- hot water
When operators apply chemical sterilization to package interiors or closures, they must remove residual chemicals before filling. This step ensures compliance with FDA regulations, maintains sensory quality, and prevents flavor degradation.
Hydrogen Peroxide Measurement, Monitoring and Control
In 21 CFR 178.1005 (a) of the Code of Federal Regulations, hydrogen peroxide is defined to be a 35% aqueous solution. In subsection (d) of this same standard, it specifies limits on the hydrogen peroxide residual. “No use of hydrogen peroxide solution in the sterilization of food packaging material shall be considered to be in compliance if more than 0.5 part per million of hydrogen peroxide can be determined in distilled water packaged under production conditions (assay to be performed immediately after packaging).”

Analytical Tools for the Detection of Hydrogen Peroxide
The traditional laboratory bench method used to determine hydrogen peroxide levels is a titration with potassium permanganate (KMnO4). This requires volumetric glassware, use of buret, and standardization of the KMnO4 prior to testing. Users typically repeat titrations up to three times to determine an averaged test result. In the manufacturing arena, where it is not uncommon for lines to produce hundreds of bottles per minute. Waiting for a lab result to confirm residuals is costly.
Paper hydrogen peroxide test strips offer advantages over titrimetric methods. Typically, the user dips a strip in a sample for a specified time and then removes it. They allow it to stand while the reagent pad develops a color reaction. The user then matches the developed color to a printed color standard. Moisture deactivates paper test strips, which is one disadvantage. Therefore, users must take care to prevent exposing the strips to air.
Even under ideal conditions, hydrogen peroxide test strips have a limited shelf-life. Furthermore, test strips may not offer the sub-ppm sensitivity required for residual testing. Lastly, user technique may influence test results. For example, how vigorously the user stirs the strip in the sample and how much they allow the sample to drain from the strip after removing it.
About CHEMetrics
CHEMetrics manufactures an innovative, colorimetric hydrogen peroxide test kit that offers great value at an economical price:
- immediate test results (in less than 2 minutes) at the point of testing, not in the lab
- long term reagent stability
- sub ppm sensitivity, and
- accuracy independent of user technique.
In this analytical system, the hydrogen peroxide liquid reagent is pre-dosed and packaged in a vacuum-sealed ampoule. In the visual test kit, the CHEMets® ampoule tip is immersed in the sample, the tip is snapped off, and the sample is automatically drawn into the ampoule. The user inverts the ampoule several times to facilitate mixing. Then, they compare it to color standard ampoules to obtain a test result. An instrumental version of this hydrogen peroxide test kit is also available. The same test procedure is followed except that the ampoule (Vacu-vials®) is read in a photometer rather than compared visually to color standards.
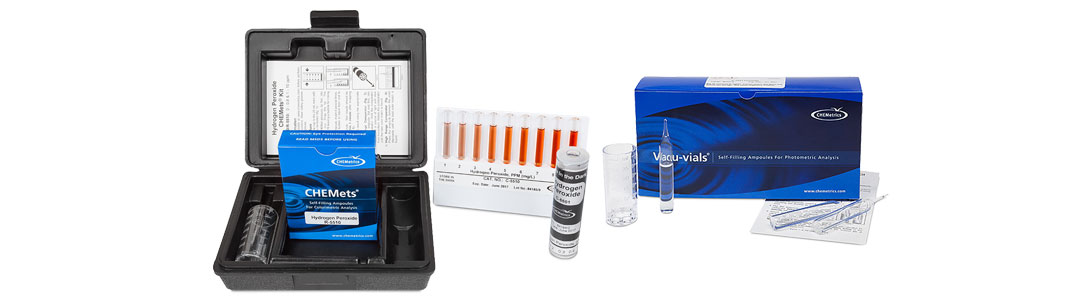
CHEMetrics Hydrogen Peroxide Test Kits
CHEMetrics offers a variety of methods and test kit configurations that permit the measurement of hydrogen peroxide concentrations ranging from sub ppm to percent levels. Food and beverage customers most widely use the ferric thiocyanate chemistry as the analytical method for hydrogen peroxide food safety. In this chemistry, the ampoules contain a ferrous ammonium thiocyanate reagent. Hydrogen peroxide in the sample converts ferrous ammonium thiocyanate to ferric thiocyanate. The intensity of the orange-brown colored ferric thiocyanate is proportional to the hydrogen peroxide level in the sample. The test delivers results in two minutes or less. The measurement range for the ferric thiocyanate CHEMets® test kit (visual) is 0 – 0.8 ppm and 1 – 10 ppm, with a detection limit of 0.05 ppm. The measurement range for the ferric thiocyanate Vacu-vials® test kit (instrumental) is 0.15 – 6.00 ppm, with a detection limit of 0.15 ppm. VACUettes® kits are also available in which the ampoules have been fitted with an auto-dilutor tip, thus allowing for measurement up to 1.2% (12,000 ppm) hydrogen peroxide.
Other CHEMetrics kit options available for measuring high levels of hydrogen peroxide (up to 20%) employ a titrimetric ceric sulfate reagent and a ferroin indicator. Titrets® ampoules employ a reverse titration method that employs pre-dosed, vacuum-sealed reagent. The user draws the sample into the ampoule in small doses until a sharp endpoint color change signals that they have reached the equivalence point. Quantitative test results are read directly from a scale printed on the side of the Titrets® ampoule.
Lastly, a ceric sulfate Go-No-Go test kit format is available upon request for situations where a Pass/Fail result at a specified control point is sufficient. The user adds a single, small dose of sample to a screw cap vial. The vial contains the hydrogen peroxide liquid reagent and endpoint indicator. An immediate color change occurs. This signals that the hydrogen peroxide level in the sample is either above or below the specified control point.
Conclusion
Food processors must weigh analysis cost, turn-around time, accuracy, sensitivity and ease of use when determining which analytical sterilization test method suits their requirements. CHEMetrics hydrogen peroxide test kits, with their “snap and read” approach to sample analysis, fulfill each of these requirements with distinction.

